Completing the Analytical Service Request Form
Fenton Woods Laboratories requires all clients to complete and submit a service request form for all analytical services. Our team uses these forms as an initial record entry into our internal workflows and as a formal document to reference for sample handling, analysis, reporting, and general sample management. This page serves as a tool to help clarify all fields in the service request form and the information we need to process your submission. All questions should be addressed to: info@fentonwoodsconsulting.com, and a member of our team will respond to your questions as soon as possible.
Name: Please enter the first and last name of the company representative Fenton Woods Laboratories should correspond with regarding sample submission, data return, sample return, and billing.
Company: Please enter the name of the company (no abbreviations).
Email: Please enter the email address of the company representative Fenton Woods Laboratories should correspond with regarding sample submission, data return, sample return, and billing.
Phone: Please enter the phone number of the company representative Fenton Woods Laboratories should correspond with regarding sample submission, data return, sample return, and billing.
Requested Service: Please enter the requested analytical service. This entry field specifies the general analytical technique requested without denoting specific instruments and techniques. For example, a company submitting drinking water for PFAS/PFOS analysis and a company submitting proteomics samples will both select LCMS; however, the former will specify QQQ LCMS analysis in the ‘Specific Analytical Service’ field whereas the latter will specify Orbitrap LCMS in the mentioned field. For any clarification on selecting the requested service, please ask one of our team members at: info@fentonwoodsconsulting.com.
Description of Sample Matrix: Describe the sample matrix in detail. Biological sample submissions should be supplemented with species information (plasma - human). Samples submitted in buffers should have a clear description of general buffer components and any additional pertinent matrix components. It is best practice to provide sufficient detail in this section so our team can evaluate the best analytical approach for your sample set.
Specific Analytical Service: Please specify the type of analytical instrument needed for your analysis. For example, if the requested service is ‘LCMS’, please specify which type of LCMS is needed (QQQ, QTOF, Orbitrap, SQ, etc). Additionally, if HPLC/GC is selected, please specify the detector type. If this field is left blank, a member of our team will reach out to you before analyzing any samples.
Study Name: Please specify the study name for the submission. Our team uses the study name for all reporting and billing purposes.
Sample Storage Condition: Please specify the sample storage condition. If unknown, please reach out to a Fenton Woods scientist ahead of time to help select the appropriate storage condition for your samples.
Description of Storage Container: Fenton Woods Laboratories prefers all HPLC and LCMS samples to be submitted in micronic HDPP vials that are oriented in a 96-well plate. Fenton Woods will accept liquid samples submitted in flip-top microcentrifuge tubes, vials, and all other closed vessels. Liquid, solid, and gas samples should be submitted in the appropriate container vessel to protect the sample and prevent spillage, evaporation, and non-specific binding. Any questions regarding best sample container practices can be directed towards our team members at: info@fentonwoodsconsulting.com.
Plate Barcode: If submitting a plate with a pre-stamped barcode, please enter the full barcode in this field. Fenton Woods will label the submitted samples with our own barcode label for internal tracking throughout the analytical workflow.
Store and Return Samples After Analysis?: Please state if Fenton Woods should return the samples to your company after completing the analysis.
Requested Completion: Please enter a requested completion date. Depending on the type of service and analysis, Fenton Woods cannot guarantee the requested completion date, and our team will try and finish the work as soon as possible.
Analytical Regulatory Domain: Please specify the regulatory domain the samples belong to. If unknown, please ask one of our team members at: info@fentonwoodsconsulting.com.
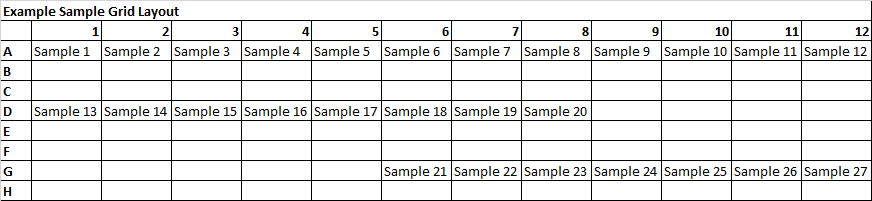
File Upload: Fenton Woods requires that all sample submissions are supplemented with the necessary corresponding documentation. For GLP and GCP sample submissions involving regulated studies, our team will follow up and request additional fields and required documentation. For all sample submissions, Fenton Woods requires a sample plate map drawn in excel. To elaborate, samples submitted in a 96 well plate must have a supplemental plate map in excel locating the sample names in the sample grid. An example of this is shown below. Additionally, customers who submit samples in cry boxes, which have a 9x9, 10x10, or another variant format must submit an excel document with all sample names in this grid. For customers who are shipping directly from a clinical or pre-clinical testing site, we ask the testing site provide this document with submission. Customers who are submitting loose samples that are not in a sample organizer must submit an excel document with the sample names. Loose samples are encouraged to be labeled with a no-fade label and clearly identifiable. Fenton Woods does not require micronic and Thermo Matrix samples to be labeled; however, our team strongly encourages that all samples submitted in these formats are affixed with the proper labels incase these samples become loose.e 27 H